Effects of Active vs. Passive Recovery in Bench Press
*Corresponding author: Luis A. Berlanga luis.berlanga@ufv.es
Cite this article
Berlanga, L.A., Matos-Duarte, M. & López-Chicharro, J. (2021). Effects of Active vs. Passive Recovery in Bench Press. Apunts Educación Física y Deportes, 145, 17-24. https://doi.org/10.5672/apunts.2014-0983.es.(2021/3).145.03
Abstract
The recovery between sets in strength exercises is one of the variables on which physical performance depends, and yet it has scarcely been investigated. Most of the studies on this topic have focused on different recovery intervals, namely the duration variable; however, our objective is to analyse whether active recovery could minimise the loss of power compared to traditional passive recovery, keeping duration constant between both protocols. To achieve this, 14 trained young volunteers did two sets of bench press of eight repetitions each and a third set until muscle failure with an optimal load to develop maximum power. Each set was separated by two minutes of passive recovery without activity or by active recovery with 60 seconds of vertical chest presses at slow speed with low load. The intraset loss of power was lower with active recovery than with the passive recovery, and this difference was statistically significant in the first and third sets (13.34 % vs. 18.84 %, p = .006; and 13.38 % vs. 17.53 %, p = .001, respectively). We also found a discreet yet significantly higher perceived exertion in the second set (4.5 vs. 5.0, p = .033). In conclusion, active recovery may be an appropriate stimulus to minimise intrasession loss of performance and improve perceived exertion in strength exercises.
Introduction
Among the variables we can control when designing strength training programmes, recovery between sets has barely been studied in the scholarly literature (Hernández-Davó et al., 2016). We know that doing more than one set during strength training may be more effective for achieving the desired goals by boosting the volume of training (Schoenfeld, 2016), hence recovery between sets is a key parameter to be borne in mind when prescribing any exercise programme and warrants greater attention for the purpose of determining optimal prescription (de Salles et al., 2009).
Although it is true that some studies have analysed different types of recovery between sets in strength training, they have focused primarily on evaluating how different time intervals influence physiological and/or performance parameters in order to determine the optimal recovery between sets depending on the objectives (Abdessemed et al., 1999; Henselmans and Schoenfeld, 2014; Hernández-Davó et al., 2017; Hernández-Davó et al., 2016; Martorelli et al., 2015; Senna et al., 2016; Willardson, 2006). These studies have focused on the variable length of the recovery period between sets. Moreover, there is considerable heterogeneity in these studies, not only in the samples (men and women, age difference, different physical fitness levels, etc.), but also in the parameters evaluated (maximum number of repetitions the person is capable of completing, percentage of loss of speed, output power, blood lactate concentration, etc.). In this regard, we believe that in the context of physical performance, investigating different types of recovery between sets in strength training should focus on evaluating its effects on the participants’ output power.
The maximum power output depends primarily on the metabolic pathways that occur in the skeletal muscle cell cytoplasm, classically known as anaerobic, particularly the phosphagen system (ATP and phosphocreatine, PCr) (Mougios, 2020). Therefore, recovery between sets should allow for a complete or almost complete resynthesis of this system in order to deliver the maximum possible performance during subsequent sets. After a brief, intense effort, such as a strength training set, 50% of initial PCr levels can be restored in the first 30 seconds of recovery and up to 90% can be resynthesised within two minutes (Chicharro and Fernández-Vaquero, 2018). PCr is synthesised thanks to the aerobic metabolic pathways, so oxygen is needed to restore the phosphagen system used during the effort. Therefore, by using active recovery between sets in strength exercises, we can facilitate the irrigation of the musculoskeletal tissue in order to improve oxygen input into the muscle cells, which may in turn help to restore the phosphagen system. This could improve performance in subsequent strength training sets (Gill et al., 2006; Latella et al., 2019).
Several authors have investigated how the length of recovery intervals affects the power output in subsequent sets during strength exercises (Hernández-Davó et al., 2016). It is suggested that for training muscle power, the rest intervals should be between 2 and 5 minutes depending on the type of exercise or the effort made (Willardson, 2006). The decision should be taken with the objective of achieving maximum if not complete resynthesis of the phosphagen system as quickly as possible. Otherwise, in subsequent sets, cytosolic glycolysis would be the predominant metabolic pathway that would satisfy the energy demand involved in the effort, with the consequent accumulation of metabolites (mainly lactate and H+ ions), which could lead to the premature appearance of muscle fatigue, in addition to a slower energy input than with the phosphagen system, taking the energy power or metabolic rate of both systems into account (amount of energy synthesised per time unit) (Mougios, 2020).
For this reason, the objective of this study was to compare the effects of active versus passive recovery on the loss of power and perceived exertion in successive sets during a strength exercise.
Methodology
Our study was a randomised crossover trial in which the participants did both types of interventions proposed: active recovery and passive recovery. Each participant visited our laboratory three times. On the first visit, they performed a maximum power test (Pmax) in the bench press, followed by a test of one maximum repetition (1MR) in the vertical chest press. The objectives of the study and the procedure were explained to them and each participant’s demographic data were recorded.
All participants signed an informed consent form and the data were processed in accordance with the laws in force as stipulated in Organic Law 15/1999 on personal data protection and in Royal Decree 1720/2007, as well as the principles enshrined in the Declaration of Helsinki (Association, 2013). This study received a favourable evaluation from the Research Ethics Committee of the Universidad Francisco de Vitoria (42/2018), where it was conducted.
Participants
Fourteen male students participated in this study. The sample size was calculated using the G*Power 3.1.9.2 software with the group of t-tests and the difference between two dependent means for paired samples according to the statistical tests for related samples (Faul et al., 2007), considering a one-tailed hypothesis test, an α probability of error of .05, a power (1-β probability of error) of .80 and an effect size of .80 (Cohen, 1992). Thus, the resulting total size was 12 participants to achieve a power of .828, so we recruited 14 volunteers, considering a possible 15% loss during the study.
The inclusion criteria were: males aged between 18 and 24 with at least one year of experience in strength training who regularly engaged in strength training (at least two days a week), were capable of lifting at least 80% of their body weight in the bench press and had no contraindication for doing physical exercise. The exclusion criteria were failure to meet any of the inclusion criteria or not being available to come to our laboratory on the scheduled days.
All the participants were encouraged to keep up their daily habits in terms of physical training and hydration and dietary patterns and were asked to refrain from training their upper body at least 48 hours before the measurement days and from consuming caffeine or any other stimulant or ergogenic aid at least three hours before the measurements were taken.
Procedures
The participants were recruited at the Universidad Francisco de Vitoria, primarily among the students in the bachelor’s degrees in Physiotherapy and Physical Activity and Sport Sciences. Potential candidates were asked to fill out on an online form with questions about their age, strength training experience, current exercise habits and possible contraindication for physical exercise. Based on the responses, the results were filtered to recruit the participants that met the inclusion criteria.
All the participants visited our laboratory three times. On the first visit, they did a guided Pmax test in the bench press, following the protocol described by other authors (Bevan et al., 2010; da Silva et al., 2015), and a 1MR test in the vertical chess press. The session started with a general warm-up comprised of five minutes of moderate-intensity cardiovascular activity and general mobility of the joints involved in the bench press exercise, followed by 3-5 minutes of passive recovery. This was followed by a specific warm-up consisting of a set of 10 repetitions of the guided bench press on a rack (Evolution Deluxe Smith Machine and Rack; Titanium Strength, S.L., Spain) without an additional load (the bar weighs 21 kg) at a controlled execution speed (two seconds in the concentric phase and two seconds in the eccentric phase) followed by 4-5 minutes of passive recovery. They subsequently performed a set of three repetitions with 20% of the 1MR estimated by the participant at the maximum execution speed possible, followed by 4-5 minutes of passive recovery.
After this warm-up, the Pmax test was executed via sets of three repetitions at the maximum speed possible with 30%, 40%, 50% and 60% of the estimated 1MR, with a passive recovery of 4-5 minutes between sets. At the end of this test, the 1MR test in the vertical chest press was performed to ascertain the load that would be used in the active recovery protocol. This 1MR test in the vertical chest press involved executing a set until muscle failure with the load equivalent to the 1MR estimated by the participant for that exercise, completing a total of 3-5 maximum repetitions and calculating the 1MR following Brzycki’s formula (Brzycki, 1993).
Approximately 7 and 14 days after the first visit to the laboratory, the volunteers participated in the two interventions to compare the differences between active and passive recovery. In a random order, the participants did one of two of the following protocols after the same general and specific warm-up that they had done on the first day. In the passive recovery protocol (PAS), two sets of eight repetitions were performed at the maximum execution speed possible with the optimal load calculated for the Pmax (OptLoad Pmax) and a third set until muscle failure, with a two-minute passive recovery between the sets (Figure 1). In the active recovery protocol (ACT), the participants performed two sets of eight repetitions at the maximum execution speed possible with the OptLoad Pmax and a third set until muscle failure, with the sets separated by a two-minute active recovery of vertical chest presses with 5%-10% of the 1MR at an execution speed of two seconds in the concentric phase and two seconds in the eccentric phase, controlled by a metronome (Metronome Beats 5.0.1) (Figure 2).
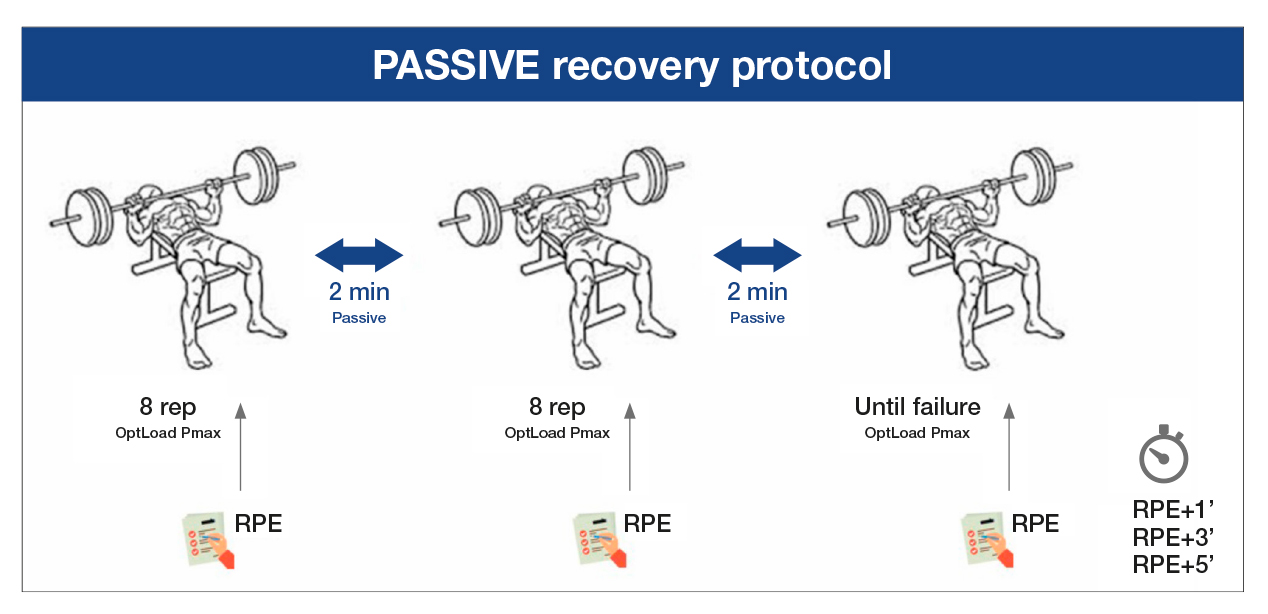
Source: Authors. Note. OptLoad Pmax: optimal load to develop maximum power; RPE: rate of perceived exertion.
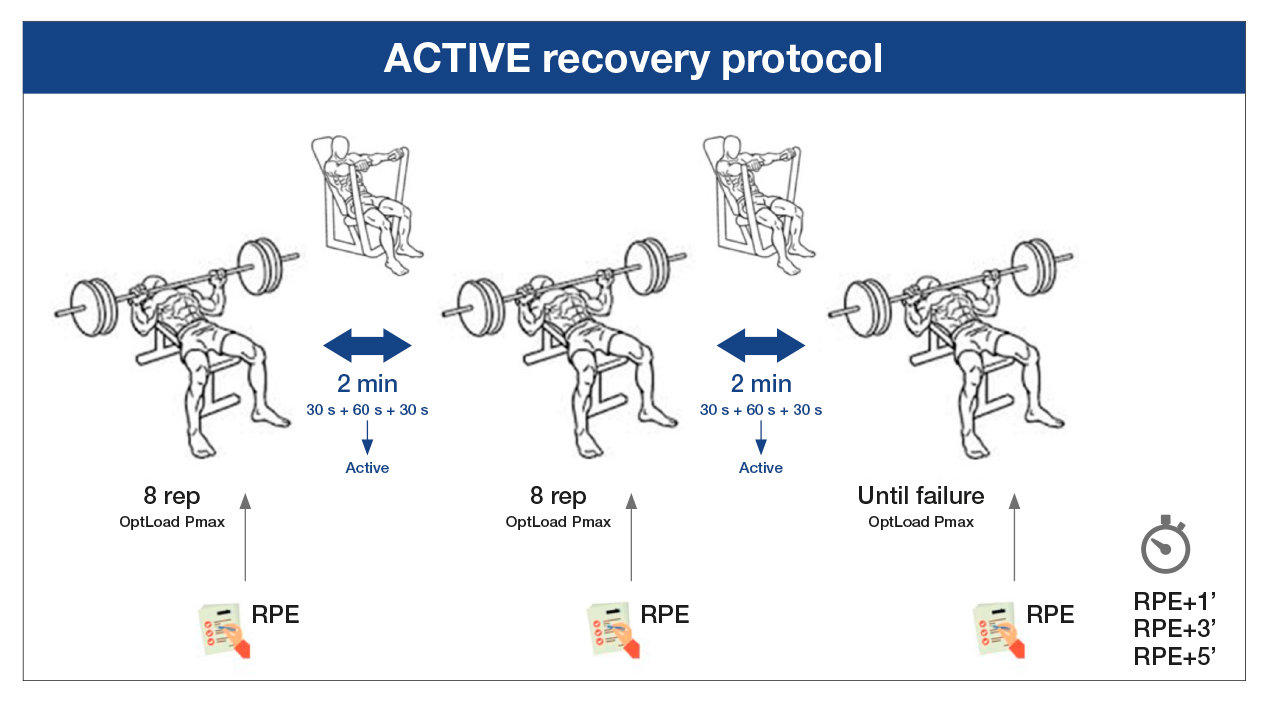
Source: Authors. Note. OptLoad Pmax: optimal load to develop maximum power; RPE: rate of perceived exertion.
Kinetic variables
The mean propulsive power (MPP) of each repetition was recorded with a linear encoder (Chronojump) at a sampling frequency of 1000Hz and software to analyse the data (Chronojump 1.8.1-95) validated by Buscà and Font (2011). The loss of the mean propulsive power (%Loss) was calculated as the difference between the MPP on the first and eighth repetition in each set, according to the calculations published by Sánchez-Medina and González-Badillo (2011) for evaluating loss of speed in strength training. The maximum number of repetitions in the third set (nMR) was recorded as the total number of repetitions performed by the participant until muscle failure.
Perceived exertion
The rate of perceived exertion (RPE) was recorded on a scale of 0 to 10 adapted for strength exercises, with a precision of .5 points allowed for the volunteers’ responses. This was recorded at the end of each set (RPE 1, RPE 2 and RPE 3, respectively) and 1, 3 and 5 minutes after the last set until muscle failure (RPE post 1’, RPE post 3’ and RPE post 5’, respectively).
Statistical analysis
All the data were analysed using the SPSS 20 statistical software (SPSS Inc., Chicago, IL, USA). The normality of each variable was verified using the Shapiro-Wilk test. A repeated-measures t-test was conducted to analyse the changes in the dependent variables associated with each protocol (ACT vs. PAS): mean propulsive power of the set (MPP, intraset loss of mean propulsive power rate (%Loss) and rate of perceived exertion (RPE). Statistical significance was set at a value of p ≤ .05 with a confidence interval of 95%.
Results
All the data in the sample presented a normal distribution in terms of age, height, weight, body mass index (BMI), strength training experience and Pmax (Table 1).

Table 1
Characteristics of the sample.
There were no significant differences in the MPP of each set between the two interventions (Table 2).

Table 2
Mean propulsive power (W) of each set in both interventions.
The intraset loss of MPP (%Loss) was lower in ACT than in PAS in all three sets (13.34% vs. 18.84%, 15.97% vs. 17.67% and 13.38% vs. 17.53%, respectively), and these differences were statistically significant for the first and third sets (p = .006 and p = .001, respectively; p = .084 for the second set) (Figure 3).
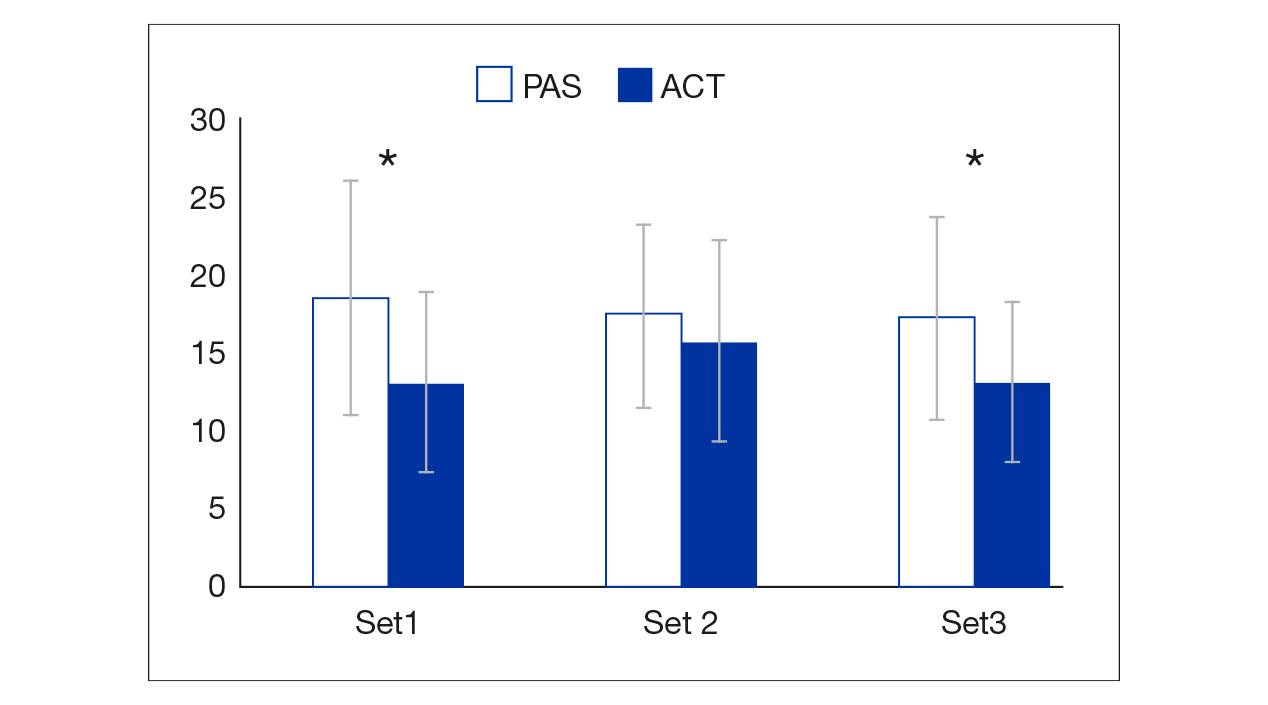
Note. ACT: active recovery protocol; PAS: passive recovery protocol.
There were no significant differences between both interventions in the nMR (45.7 ± 11.9 for the ACT protocol vs. 45.6 ± 11.6 for the PAS protocol).
The RPE was virtually the same in both protocols, although the result of the second set was significantly higher in ACT than in PAS (4.5 vs. 5.0, p = .033). Similarly, during the recovery after both protocols, the RPE tended to be slightly lower in ACT than in PAS during the measurements after 1, 3 and 5 minutes (9.2 vs. 9.3, 4.8 vs. 5.1 and 3.2 vs. 3.4, respectively), although these differences were not statistically significant (Figure 4).
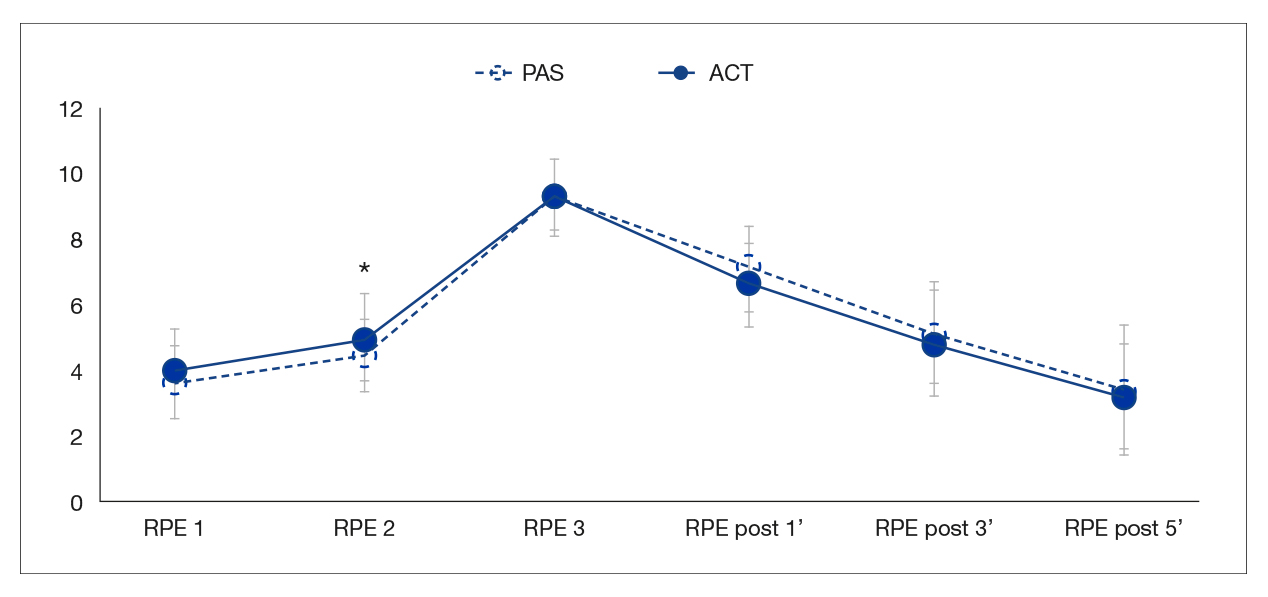
Note. ACT: active recovery protocol; PAS: passive recovery protocol; RPE: rate of perceived exertion.
Discussion
To our knowledge, this is the first study to analyse the effects of an active recovery using the same movement as the exercise evaluated on kinetic and perceptual variables in strength exercises. One of the most noteworthy aspects of our intervention is that the active recovery was well tolerated by all the participants and did not lead to any reduction in physical performance or perceived exertion.
Although we did not find significant differences between both interventions in the MPP of each set, there were differences between the protocols when we calculated the intraset loss of power, it transpiring that the active recovery protocol reduced the loss of power between successive sets compared to passive recovery; these differences were statistically significant in the first and third sets. Thus, knowing that less creatine kinase (CK) is produced when active versus passive recovery stimuli are compared (Gill et al., 2006), and that active stimuli promote oxygen perfusion and improve the recovery of the muscles involved in a given physical effort (Latella et al., 2019), we could hypothesise that our active recovery proposal could promote muscle tissue blood irrigation, with the consequent oxygen input, in turn fostering PCr resynthesis and therefore a greater involvement of the phosphagen system in successive sets.
In this same vein, Schoenfeld et al. (2019) conducted a study analysing whether a two-minute active recovery comprised of 30 seconds of voluntary isometric contraction of the muscle group involved in the exercise followed by 90 passive seconds would improve performance and structural adaptations compared to a passive recovery of the same length (two minutes), in an intervention three times a week over an eight-week period. The results showed that active recovery achieved higher hypertrophy in the lower limbs but not in the upper limbs, although this intervention did not show significant improvements compared to passive recovery in either strength or muscle resistance, which concurs with our findings about MPP and nMR. The authors assert that the greater degree of hypertrophy of the lower limbs associated with active recovery may be due to the fact that isometric contraction may lead to local blood vessel constriction, causing metabolites to accumulate (particularly H+), which would be conducive to a positive adaptation of acidosis buffering capacity; however, they did not measure metabolic stress markers and recommend that it be measured in future studies.
Other studies have evaluated different recovery strategies between strength training sets, including the systematic review by Latella et al. (2019). These authors identified 396 studies and ultimately analysed 26 that included different active recovery strategies between sets such as stretches, aerobic exercise, massage and self-myofascial release, vibration or electrical stimulation, among others. Their conclusions affirm that including active stimuli in recovery may increase the total number of repetitions that can be done; improve kinetic variables such as strength, power and speed; increase muscle activation; and reduce perceived exertion. However, it is important to note that the heterogeneity of the studies included in this review makes it difficult to draw general conclusions as to which active stimuli might be most appropriate for each strength training session. Furthermore, none of them studied an active recovery that included the same movement as that which is performed during training, as ours did. Nonetheless, these findings may help to corroborate the benefits of active versus passive recovery in strength training.
One of these active stimuli, aerobic exercise, was analysed by Mohamad et al. (2012), who conducted a crossover study to compare the acute responses of four types of interventions on different physiological and performance parameters. Although this active stimulus is different to our proposal in this study, their results show that there were no significant differences between groups or in kinetic variables, as in our case in MPP and nMR, as neither were there any in the kinematic variables or lactate concentrations. However, as the actual authors note, other possible important benefits of active stimulus in recovery periods between strength sets were not evaluated, such as the improvement in the resynthesis of energy substrates, the increase in anabolic hormone response and the lower loss of power during active recovery compared to passive recovery.
Another possible mechanism which could explain the lower intraset loss of power found by us with the ACT protocol compared to the PAS protocol may be due to the greater motor plate excitability induced by the active stimulus, which could influence the afferent pathway related to the critical threshold of peripheral fatigue and therefore the central command, thus facilitating muscle contractibility in successive sets during strength training (Allen et al., 2008).
While our study did not evaluate physiological parameters such as motor plate excitability, lactatemia or other muscle metabolic activity indicators, we do know that intense muscle activity causes PCr levels to decrease, which increases the concentration of inorganic phosphorous in muscle cells. Furthermore, ATP turnover and the increase in cytosolic metabolic activity increase the concentration of H+ ions, particularly in type-II fibres, in which pH could drop from 7.0 to 6.2 (Kent-Braun et al., 2012). Therefore, future studies addressing the effects of active recovery during strength training should also measure physiological parameters such as blood lactate levels, muscle oxygenation and CK levels.
Conclusions
In conclusion, our results demonstrate that given the same duration, active recovery may be more effective than passive recovery as a strategy to minimise loss of power and improve perceived exertion in successive sets during a strength training exercise in trained young men.
However, one of the main limitations of our study is that we did not record, for example, physiological or other parameters related to changes in motor plate excitability or possible injury mechanisms, so we cannot discern the mechanisms associated with this possible improvement. Similarly, another limitation lies in the actual experimental design, as including more sets could have shown the differences between the two recovery protocols in successive sets during a strength training exercise more clearly, as well as the use of a subjective scale to evaluate perceived exertion.
Future avenues of research should include and compare both physiological and performance parameters and analyse the effects of active versus passive recovery on the upper and lower limbs.
References
[1] Abdessemed, D., Duche, P., Hautier, C., Poumarat, G. & Bedu, M. (1999). Effect of recovery duration on muscular power and blood lactate during the bench press exercise. Int J Sports Med, 20(6), 368-373. https://doi.org/10.1055/s-2007-971146
[2] Allen, D. G., Lamb, G. D. & Westerblad, H. (2008). Skeletal muscle fatigue: cellular mechanisms. Physiol Rev, 88(1), 287-332. https://doi.org/10.1152/physrev.00015.2007
[3] Association, W. M. (2013). World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Jama, 310(20), 2191-2194. https://doi.org/10.1001/jama.2013.281053
[4] Bevan, H. R., Bunce, P. J., Owen, N. J., Bennett, M. A., Cook, C. J., Cunningham, D. J., Newton, R. U. & Kilduff, L. P. (2010). Optimal loading for the development of peak power output in professional rugby players. J Strength Cond Res, 24(1), 43-47. https://doi.org/10.1519/JSC.0b013e3181c63c64
[5] Brzycki, M. (1993). Strength Testing – Predicting a One-Rep Max from Reps-to-Fatigue. Journal of Physical Education, Recreation & Dance, 64(1), 88-90. https://doi.org/10.1080/07303084.1993.10606684
[6] Chicharro, J. L. & Fernandez Vaquero, A. (2018). Bioenergética de las fibras musculares y ejercicio. Madrid: Exercise Physiology & Training.
[7] Cohen J. (1992). A power primer. Psychological bulletin, 112(1), 155–159. https://doi.org/10.1037//0033-2909.112.1.155
[8] da Silva, B. V., Simim, M. A., Marocolo, M., Franchini, E. & da Mota, G. R. (2015). Optimal load for the peak power and maximal strength of the upper body in Brazilian Jiu-Jitsu athletes. J Strength Cond Res, 29(6), 1616-1621. https://doi.org/10.1519/jsc.0000000000000799
[9] de Salles, B. F., Simao, R., Miranda, F., Novaes Jda, S., Lemos, A. & Willardson, J. M. (2009). Rest interval between sets in strength training. Sports Med, 39(9), 765-777. https://doi.org/10.2165/11315230-000000000-00000
[10] Faul, F., Erdfelder, E., Lang, A.G., & Buchner, A. (2007). G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behavior Research Methods 39(2), 175-191. http://doi.org/10.3758/BF03193146
[11] Gill, N. D., Beaven, C. M. & Cook, C. (2006). Effectiveness of post-match recovery strategies in rugby players. Br J Sports Med, 40(3), 260-263. http://dx.doi.org/10.1136/bjsm.2005.022483
[12] Henselmans, M. & Schoenfeld, B. J. (2014). The effect of inter-set rest intervals on resistance exercise-induced muscle hypertrophy. Sports Med, 44(12), 1635-1643. https://doi.org/10.1007/s40279-014-0228-0
[13] Hernández-Davó, J. L., Botella Ruiz, J. & Sabido, R. (2017). Influence of strength level on the rest interval required during an upper-body power training session. J Strength Cond Res, 31(2), 339-347. https://doi.org/10.1519/jsc.0000000000001512
[14] Hernández-Davó, J. L., Solana, R. S., Sarabia Marín, J. M., Fernández Fernández, J. & Moya Ramón, M. (2016). Rest interval required for power training with power load in the bench press throw exercise. J Strength Cond Res, 30(5), 1265-1274. https://doi.org/10.1519/jsc.0000000000001214
[15] Kent-Braun, J. A., Fitts, R. H. & Christie, A. (2012). Skeletal muscle fatigue. Compr Physiol, 2(2), 997-1044. https://doi.org/10.1002/cphy.c110029
[16] Latella, C., Grgic, J. & Van der Westhuizen, D. (2019). Effect of interset strategies on acute resistance training performance and physiological responses: a systematic review. J Strength Cond Res, 33, Suppl 1, S180-s193. https://doi.org/10.1519/jsc.0000000000003120
[17] Martorelli, A., Bottaro, M., Vieira, A., Rocha-Junior, V., Cadore, E., Prestes, J., Wagner, D. & Martorelli, S. (2015). Neuromuscular and blood lactate responses to squat power training with different rest intervals between sets. J Sports Sci Med, 14(2), 269-275
[18] Mohamad, N. I., Cronin, J. B. & Nosaka, K. K. (2012). The effect of aerobic exercise during the interset rest periods on kinematics, kinetics, and lactate clearance of two resistance loading schemes. J Strength Cond Res, 26(1), 73-79. https://doi.org/10.1519/JSC.0b013e31821bf1f5
[19] Mougios, V. (2020). Exercise Biochemistry (2nd Edition). Human Kinetics.
[20] Sánchez-Medina, L., & Gonzalez-Badillo, J. J. (2011). Velocity loss as an indicator of neuromuscular fatigue during resistance training. Med Sci Sports Exerc, 43(9),1725-1734. https://doi.org/10.1249/MSS.0b013e318213f880
[21] Schoenfeld, B. (2016). Science and development of muscle hypertrophy. Champaign, IL: Human Kinetics.
[22] Schoenfeld, B. J., Grgic, J., Contreras, B., Delcastillo, K., Alto, A., Haun, C., O De Souza, E. & Vigotsky, A. D. (2019). To flex or rest: does adding no-load isometric actions to the inter-set rest period in resistance training enhance muscular adaptations? A randomized-controlled trial. Front Physiol, 10, 1571. https://doi.org/10.3389/fphys.2019.01571
[23] Senna, G. W., Willardson, J. M., Scudese, E., Simao, R., Queiroz, C., Avelar, R. & Martin Dantas, E. H. (2016). Effect of different interset rest intervals on performance of single and multijoint exercises with near-maximal loads. J Strength Cond Res, 30(3), 710-716. https://doi.org/10.1519/jsc.0000000000001142
[24] Willardson, J. M. (2006). A brief review: factors affecting the length of the rest interval between resistance exercise sets. J Strength Cond Res, 20(4), 978-984. https://doi.org/10.1519/R-17995.1
ISSN: 2014-0983
Received: November 23, 2020
Accepted: March 30, 2021
Published: July 01, 2021
Editor: © Generalitat de Catalunya Departament de la Presidència Institut Nacional d’Educació Física de Catalunya (INEFC)
© Copyright Generalitat de Catalunya (INEFC). This article is available from url https://www.revista-apunts.com/. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit https://creativecommons.org/licenses/by-nc-nd/4.0/deed.en


